VIPUN Medical and KU Leuven announce the full enrolment of the ANTERO-2 clinical study, evaluating the safety and feasibility of using the VIPUN Gastric Monitoring System in the Intensive Care Unit, and the ANTERO-3 clinical study, evaluating the impact of enteral nutrition infusion rate on gastric motility, measured with the VIPUN Gastric Monitoring System.
VIPUN Medical bvba, a KU Leuven spin-off aiming to improve clinical outcomes by developing medical devices for stomach function monitoring, and the University of Leuven, announce that all study subjects have been enrolled and completed all visits in its ANTERO-2 and the ANTERO-3 clinical studies.
The VIPUN Gastric Monitoring System (GMS) aims to provide medical nutrition guidance to Intensive Care Unit (ICU) and post-operative hospital staff to reduce enteral feeding intolerance (EFI). A total of 30 Intensive Care Patients (ANTERO-2) and 12 healthy subjects (ANTERO-3) were enrolled and completed all visits.
The purpose of the ANTERO-2 in-patient pilot study is to evaluate the safety and feasibility of using the VIPUN GMS in the Intensive Care Unit, as well as studying the relationship between gastric motility (as measured by the VIPUN GMS) and gastric emptying. The purpose of ANTERO-3 study is to evaluate the effect of different intragastric infusion rates of enteral nutrition on gastric motility, emptying and their correlation (NCT03664570).
Positive data from an earlier study with the VIPUN GMS (ANTERO-1) have previously demonstrated the safety and feasibility of gastric motility measurement. Moreover, the study showed that pharmacologically-delayed gastric emptying was clearly linked with decreased gastric motility as measured and quantified by the VIPUN GMS (n=18), showing the ability of the system to pick up clinically significant reduction in gastro-intestinal function.
EFI in the ICU and post-operative ward is the result of medication such as opioids, inflammation, surgery, electrolyte imbalances, hyperglycemia, sepsis, increased cranial pressure, impaired function of the enteric nerves and smooth muscles of the GI tract in general and the presence of the underlying disease/trauma itself and is associated with complications ranging from mild and frequent (diarrhea, nausea, abdominal discomfort) over moderate [(ventilator associated) aspiration pneumonia] to severe and rare (intestinal ischemia, septic shock, and ultimately death).
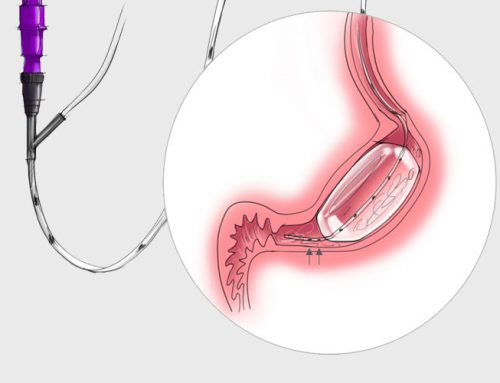
The VIPUN GMS consists of a balloon mounted on a dual lumen naso-gastric feeding tube and a monitoring unit with embedded interpretation algorithm. Through accurate monitoring of gastric motility in the ICU and post-operative ward, VIPUN Medical plans to offer physicians an objective measure of gastro-intestinal function leading to individualized medical nutrition care. As a result it is expected that the incidence and impact of enteral feeding-related complications decreases while increasing nutrition success.
Prof Jan Tack, MD, PhD, principal investigator of the ANTERO-2 and 3 studies, comments: “We are delighted to announce the completion of our second and third clinical study evaluating the safety and functionality of the VIPUN GMS for the monitoring of stomach motility. These studies provide important insights into the use of the VIPUN GMS in the ICU as well as the complex neurohumoral feedback mechanism which regulates gastric emptying of enteral nutrition through changes in gastric motility. We believe this can be an important step in determining optimal enteral nutrition strategies and infusion speed depending on tolerance level of individual patients”.
Nico Van Tichelen, CEO of VIPUN Medical bvba, comments: “We believe the VIPUN GMS will provide an objective parameter to physicians for medical nutrition management, increasing success of medical nutrition and reducing feeding related complications in ICU and post-operative patients. We look forward to the ANTERO-2 and ANTERO-3 study conclusions as well as the start of the ANTERO-4 study evaluating the effect of a gastroprokinetic medication on the VIPUN GMS monitoring read-out.